 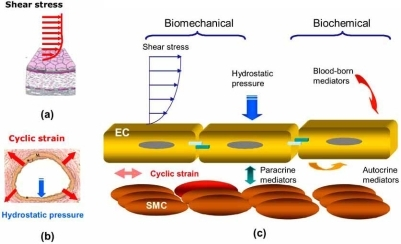
Here, CREB activation leads to the transcription of a set of CRE-regulated genes that overlaps, but is distinct from, the genes expressed following L-type Ca 2+ channel activation 10, 14. Ca 2+ influx in response to depletion of the sarcoplasmic reticulum (SR) Ca 2+ stores (store-operated Ca 2+ entry, SOCE) also activates CREB in both cultured VSMCs and intact arteries. In agreement with this, cerebral arteries from hypertensive rats are depolarized compared to normotensive animals and have elevated cytosolic Ca 2+ and increased CREB phosphorylation/c-fos expression, which can be reversed by L-type Ca 2+ channel inhibition 13. Voltage-dependent Ca 2+ influx through L-type Ca 2+ channels induces CREB phosphorylation in both cultured VSMCs and in intact mouse cerebral arteries 11, 12 and correlates with an increase in transcription of proliferation-associated CRE-containing genes such as c-fos and early growth response-1, egr-1 10, 11, 12. Intriguingly, Ca 2+ signals from different sources trigger different patterns of CRE-regulated VSM gene expression 10. Ser 133 is a substrate for a number of kinases that are either directly or indirectly activated by Ca 2+, including protein kinase A (PKA), protein kinase C, members of the calcium/calmodulin-dependent kinase (CaMK) family and ribosomal S6 kinase (RSK) −2 6. Multiple Ca 2+-regulated signaling cascades converge to phosphorylate CREB at a critical Ser 133 residue, which induces recruitment of coactivators and the formation of an active transcriptional complex 9. CREB binds as a dimer to a conserved cyclic AMP response element (CRE) in the promoter of pro-survival genes and activates transcription in response to a variety of diverse extracellular signals 8. The altered pattern of gene expression that drives changes in VSMC phenotype correlates with different patterns of Ca 2+ signaling 6, 7, although the molecular basis of Ca 2+-regulated gene expression in VSMCs is not well understood.Ĭa 2+-induced gene expression can be mediated in VSMCs by the transcription factor Ca 2+/cyclic AMP response element binding protein (CREB) 6. Proliferation of VSMCs in the vessel wall contributes to atherogenesis, vascular remodelling in pulmonary hypertension, and to restenosis, a narrowing of the vessel lumen following balloon angioplasty and bypass vein grafting 3, 4, 5. This plasticity is important for vessel repair at times of vascular injury, but is also a central feature of vascular disease. 
Quiescent VSMCs change to a synthetically active and proliferative phenotype in response to stimuli such as mechanical stress, growth factors or inflammatory mediators 2. In healthy blood vessels, the majority of vascular smooth muscle cells (VSMCs) are quiescent, contractile and proliferate slowly 1. sAC, which is insensitive to G-protein modulation but responsive to Ca 2+, pH and ATP, may therefore act as an overlooked regulatory node in vascular Ca 2+-transcription coupling. This suggests that SOCE in hCASMCs activates sAC which in turn activates the cAMP/PKA/CREB axis. Transmembrane adenylyl cyclase inhibition had no effect on the SOCE-induced rise in cAMP, while sAC inhibition abolished SOCE-generated cAMP and significantly reduced SOCE-induced VASP and CREB phosphorylation. SOCE also activated the cAMP effector, protein kinase A (PKA), as determined by the PKA reporter, AKAR4-NES, and induced phosphorylation of vasodilator-stimulated phosphoprotein (VASP) and CREB. Store-operated Ca 2+ entry (SOCE) into hCASMCs expressing the FRET-based cAMP biosensor H187 induced a rise in cAMP that mirrored cytosolic. Here we show that the Ca 2+/HCO 3 −-sensitive enzyme, soluble adenylyl cyclase (sAC), links Ca 2+ influx in human coronary artery smooth muscle cells (hCASMCs) to 3′,5′-cyclic adenosine monophosphate (cAMP) generation and phosphorylation of the transcription factor Ca 2+/cAMP response element binding protein (CREB). Although not well understood this allows normally contractile VSMCs to become proliferative following vessel injury, a process essential for repair but which also contributes to vascular remodelling, atherogenesis and restenosis. Ca 2+-transcription coupling controls gene expression patterns that define vascular smooth muscle cell (VSMC) phenotype. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
